Summary of the FSRH (2023, amended 2025) guideline on Intrauterine Contraception (IUC / IUD / IUS / LNG-IUD)
Key Principles & Rationale
- IUC (both copper and levonorgestrel (LNG) types) are very effective, long-acting reversible contraceptives.
- Their contraceptive failure rate in the first year is ~0.6–0.8 %.
- Effectiveness is not reduced by enzyme-inducing drugs or increased body mass index (BMI).
- Mode of action is predominantly pre-fertilisation (inhibiting sperm/ovum interaction), though some secondary effects on the endometrium also occur.
Types and Duration of Use
The FSRH guideline recognises two main types of intrauterine contraception — the levonorgestrel-releasing intrauterine device (LNG-IUD) and the copper intrauterine device (Cu-IUD).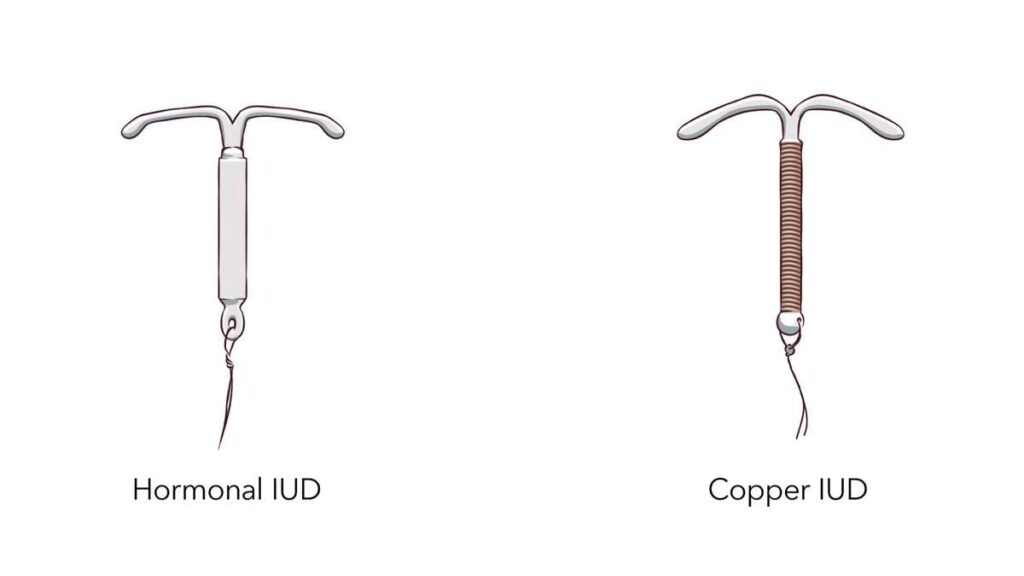
For LNG-IUDs (previously referred to as LNG-IUS), such as the 52 mg devices, FSRH supports their contraceptive use for up to 6 years under the standard licence. However, according to the latest FSRH Clinical Effectiveness Unit (CEU) statement, all 52 mg LNG-IUDs can be safely used for up to 8 years for contraception if inserted before the age of 45. When inserted at age 45 years or above, the device may be retained until the woman reaches age 55, after which contraception is no longer required. When an LNG-IUD is being used as part of hormone replacement therapy (HRT) for endometrial protection, the recommended duration of use remains 5 years only.
For Copper IUDs (Cu-IUDs), typically those with a copper surface area of at least 300 mm², the device can be used as long as needed until menopause, as there is no strict expiry if the device remains functional and the woman wishes to continue. For women aged 40 years or older at the time of insertion, FSRH states that the same device may safely remain in place until menopause, without the need for replacement.
It is also worth noting that the guideline has updated terminology: the previous distinction between IUS (for hormonal) and IUD (for non-hormonal) has been replaced by a unified term — “intrauterine device (IUD)” — with descriptive modifiers, namely LNG-IUD for hormonal devices and Cu-IUD for non-hormonal ones.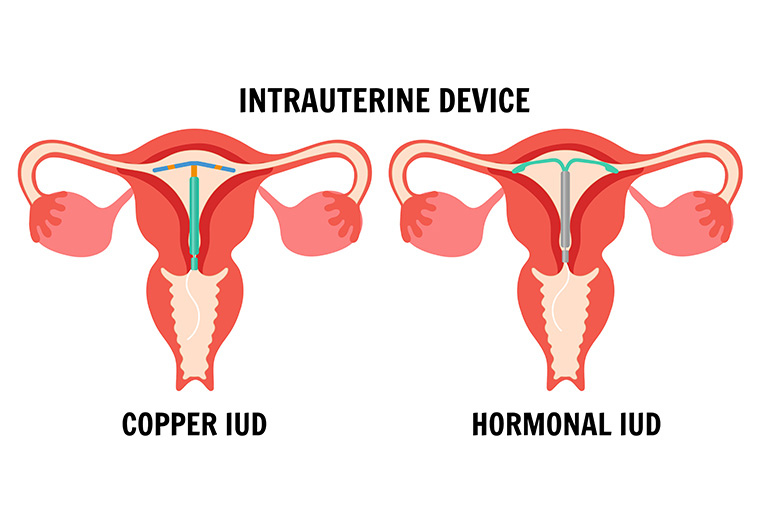
Contraindications & Medical Eligibility (UKMEC / FSRH)
FSRH uses a “UKMEC-style” categorisation for who can or cannot use IUC, adapted into this guidance.
Typical contraindications (category 4 or 3 caution) include:
- Current or recent pelvic inflammatory disease (PID), untreated cervicitis or endometritis
- Known uterine anatomical abnormalities (e.g. distortion)
- Known pregnancy or suspicion of pregnancy
- Unexplained vaginal bleeding
- Uterine or cervical cancer
- Wilson’s disease (for Cu-IUD)
- Breast cancer (for LNG-IUD) — the guideline updates recent evidence; the absolute risk remains very small. cosrh.org+1
Also caution in:
- Immunocompromise or high risk of STI — consider screening before insertion
- History of ectopic pregnancy (still allowed, but counselling needed)
- Nulliparous women: not a contraindication — IUDs are safe and acceptable, though insertion may be slightly more challenging. Society of Family Planning+2cosrh.org+2
Pre-insertion Assessment & Counselling
Essential points to cover:
- Full contraceptive counselling: effectiveness, advantages, disadvantages, side effects, and bleeding patterns (especially with LNG-IUD). cosrh.org+1
- Medical history, risk of STIs, pelvic infection history
- Pregnancy exclusion before insertion
- Informed consent including possibility of pain, bleeding, perforation, expulsion
- Use of local anaesthetic (e.g. 2.5% lidocaine + 2.5% prilocaine cream) may reduce insertion pain. cosrh.org
- Insertion should be by trained clinician, following aseptic technique. cosrh.org
There is a pre-insertion checklist in the guideline. cosrh.org
Timing of Insertion & Switching
Timing for insertion:
- If reasonably certain not pregnant, IUD can be inserted at any time in the cycle (no need to wait for menses) for Cu-IUD; for LNG-IUD, additional precautions (backup) if not inserted in first 7 days. srh.bmj.com+1
- Postpartum: immediate insertion is acceptable (within 10 minutes of placental delivery or during cesarean) provided no infection. Higher expulsion rates should be considered. srh.bmj.com+2cosrh.org+2
- Post-abortion: can be inserted immediately after abortion (unless there was infection). cosrh.org+1
Switching from another contraceptive:
- From copper IUD → LNG-IUD: can be done any time if pregnancy is excluded. Additional precaution for 7 days may be advised per switching table.
- From combined hormonal contraception (CHC): if switching during days 1–7 of active pill, insertion and no additional contraception; else may need 7 days backup.
- From progestogen-only methods / injectables: tables exist; e.g. LNG-IUD may require backup for 7 days in some scenarios.
Aftercare, Follow-up & Troubleshooting
- Routine post-insertion check is not required (except for IUDs inserted within 48 hrs postpartum).
- Users should be advised to check the threads 4–6 weeks after insertion and thereafter monthly (or after menses) to ensure device is still in place.
- If threads are not visible: speculum + possibly ultrasound or further steps (see algorithm in guideline).
- If the device is confirmed in proper uterine location, reassure and leave.
- On removal: check the device is intact and expected type.
- Difficult removal: refer to more experienced provider.
Complications and Management
Complications associated with intrauterine contraception (IUC) are uncommon, and the benefits of use far outweigh the risks.
Uterine perforation is a rare complication, occurring in fewer than 1 per 1,000 insertions. It may be suspected if the woman experiences severe pain, abnormal bleeding, or if the IUD threads are not visible. In such cases, imaging (usually pelvic ultrasound or X-ray) should be performed to confirm the device’s location, and removal should be arranged by a specialist if perforation is confirmed.
Expulsion of the device can occur, most commonly in the postpartum period, in nulliparous women, or when insertion is performed soon after delivery. If expulsion is confirmed, the device can be replaced immediately if the woman wishes to continue using this method, or she should be counselled about alternative contraception.
Bleeding irregularities are common, especially in the initial months after insertion. The copper IUD is associated with heavier and more prolonged menstrual bleeding, whereas the LNG-IUD may cause irregular bleeding initially but often leads to reduced bleeding or amenorrhoea with continued use. These symptoms are usually benign and improve over time. Management is primarily symptomatic, using nonsteroidal anti-inflammatory drugs (NSAIDs) or tranexamic acid, and other causes of abnormal bleeding should be excluded if symptoms persist.
Pain or cramping may occur during or shortly after insertion and usually settles with simple analgesia and reassurance. Persistent or worsening pain warrants evaluation for infection, partial expulsion, or perforation.
Pelvic infection or pelvic inflammatory disease (PID) risk is slightly increased in the first 20 days following insertion but remains very low overall. Routine prophylactic antibiotics are not recommended; however, women should be screened for sexually transmitted infections (STIs) prior to insertion if risk factors are present.
If pregnancy occurs with an IUD in situ, the absolute risk of ectopic pregnancy is lower than in women not using contraception, but the relative proportion of ectopic pregnancies is higher compared to intrauterine pregnancies. Any woman who becomes pregnant with an IUD in place should be assessed for possible ectopic pregnancy and managed according to standard obstetric guidelines.
Finally, fracture of the device is a rare occurrence and may complicate removal. If suspected, imaging should be undertaken to identify retained fragments, and removal should be performed by an experienced clinician, often under ultrasound guidance.
Special Populations & Considerations
- Nulliparous women: IUDs are not contraindicated and are safe; may have more discomfort or more difficult insertion. Adolescents / young women: generally appropriate with counselling
- Breastfeeding / postpartum: safe, with awareness of higher expulsion risk early postpartum
- Women approaching menopause: Cu-IUD may safely be kept until menopause; LNG-IUD may be used until age 55 if inserted before 45.
- Women needing endometrial protection on HRT: LNG-IUD devices can be used (5-year use) as part of HRT care.
- Women with bleeding disorders or on anticoagulants: the guideline has an algorithm (instrumentation in these women) and counsels risk versus benefit.
What’s New / Updates to Note (for 2023 guideline)
- The extension of 52 mg LNG-IUD use from 5/6 years to up to 8 years for contraception in users <45 years (supported by FSRH CEU) is a key change.
- Increased flexibility in nomenclature (both hormonal and non-hormonal are called IUD, with qualifiers) to align with international usage.
- For Cu-IUD in older age groups (≥40), extended use until menopause is accepted.
Complications and Management (Detailed Summary for MRCOG Part 2)
Although intrauterine contraception (IUC) is among the safest and most effective forms of reversible contraception, awareness of potential complications and their management is essential for both clinical practice and examination scenarios.
1. Uterine Perforation
Incidence:
Perforation is rare, occurring in fewer than 1 per 1,000 insertions, and typically happens at the time of insertion.
Risk factors:
Postpartum or lactating women (particularly within 36 weeks of delivery)
Uterine anomalies or extreme anteversion/retroversion
Inexperienced operator or difficult insertion
Clinical features:
Perforation may be complete (device passes through the uterine wall into the peritoneal cavity) or partial (embedded in the myometrium). It should be suspected if:
Severe or persistent pain during insertion
Unexpected bleeding
Threads are not visible on follow-up
Device not visualised on ultrasound within the uterine cavity
Management:
If perforation is suspected, perform pelvic ultrasound first. If not seen in the uterus, an abdominal X-ray can locate the device.
If the device is within the uterine wall or peritoneal cavity, laparoscopic removal is usually indicated.
The patient should be counselled and offered alternative contraception until removal.
Antibiotic prophylaxis is not routinely required unless infection is suspected.
2. Expulsion
Incidence:
Expulsion occurs in approximately 2–10 % of users within the first year, most commonly in the first few months post-insertion.
Risk factors:
Insertion soon after delivery or second-trimester abortion
Heavy or painful menstruation
Nulliparity (slightly higher risk)
Improper fundal placement
Clinical signs:
Missing or lengthened threads
Pain, cramping, or change in bleeding pattern
Feeling the device at the cervical os
Management:
If expulsion is complete, offer replacement if the woman wishes to continue IUC or counsel on alternative contraception.
If partial expulsion (device partly visible in cervix), remove and replace with a new one — the partially expelled device should not be repositioned.
Backup contraception should be used until effective replacement.
3. Bleeding Disturbances
Incidence and Patterns:
Changes in bleeding are common, especially during the first 3–6 months.
Copper IUDs often cause heavier, longer, or more painful periods.
LNG-IUDs cause irregular bleeding or spotting initially, followed by amenorrhoea or light bleeding after several months.
Mechanism:
Copper causes local inflammatory effects on the endometrium, increasing prostaglandin release, while LNG-IUD leads to endometrial suppression and reduced vascularity.
Management:
Reassure women that bleeding changes usually settle within 3–6 months.
NSAIDs (e.g., mefenamic acid, ibuprofen) or tranexamic acid may reduce heavy or painful bleeding.
Exclude other causes of abnormal bleeding (pregnancy, infection, fibroids, polyps, cervical pathology).
Persistent abnormal bleeding with LNG-IUD should prompt review for other pathology.
4. Pain and Cramping
Pain and uterine cramping are common during and shortly after insertion.
Usually mild and self-limiting.
Managed with reassurance and simple analgesia (NSAIDs or paracetamol).
If persistent or severe, assess for malposition, infection, or perforation via examination and ultrasound.
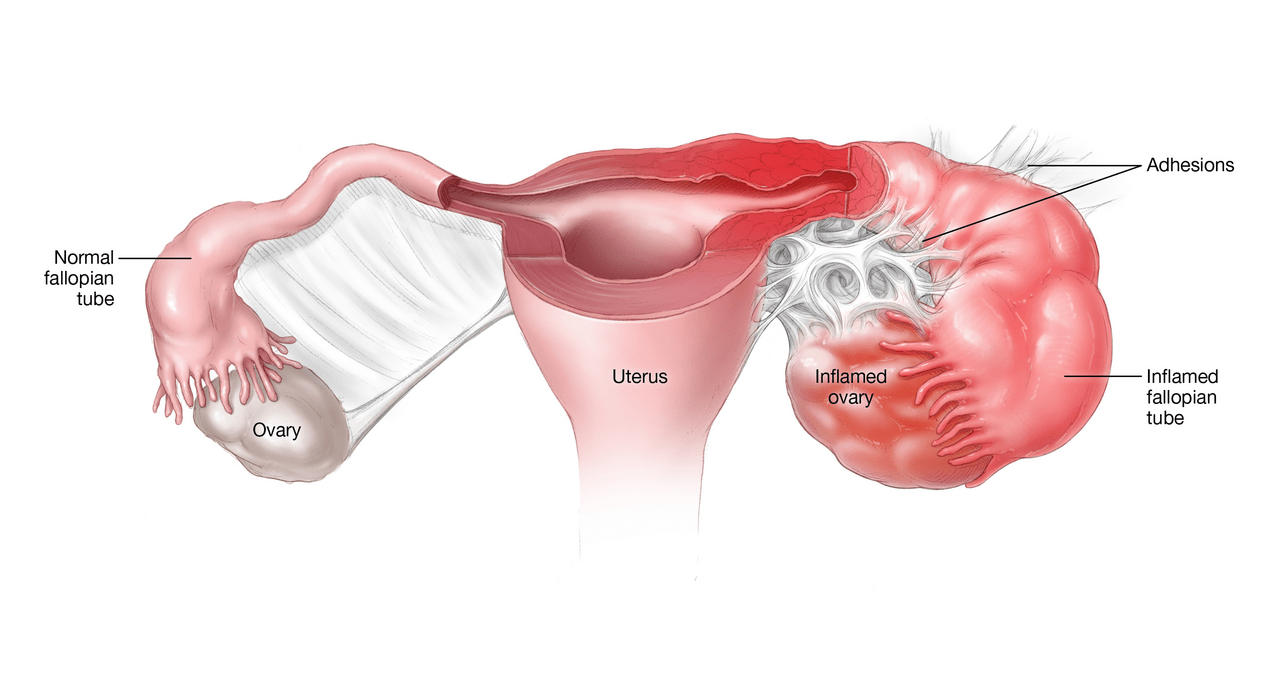
5. Pelvic Infection (PID)
Incidence:
The risk of PID is slightly increased only in the first 20 days after insertion (estimated <1%), after which the risk returns to baseline.
Pathophysiology:
This early risk is related to contamination during insertion, not to the presence of the device itself.
Risk factors:
Multiple sexual partners
Age <25 years
Previous or current STI
Lack of pre-insertion STI screening
Management:
Routine prophylactic antibiotics are NOT recommended.
Women at risk (e.g., under 25, new/multiple partners) should be screened for chlamydia and gonorrhoea before or at insertion.
If PID is suspected (pelvic pain, discharge, fever), treat with appropriate antibiotics as per NICE/FSRH infection guidance.
The IUD does not need to be removed immediately unless symptoms are severe or not improving within 72 hours of treatment.
6. Pregnancy with IUD In Situ
Incidence:
Extremely rare (<1 per 100 women-years).
Key point:
If pregnancy occurs with an IUD in place, the absolute risk of ectopic pregnancy is low, but relative risk (percentage of all pregnancies that are ectopic) is higher.
Management:
First step: Exclude ectopic pregnancy by clinical assessment and ultrasound.
If intrauterine pregnancy confirmed and woman wishes to continue:
Remove the IUD if the threads are visible (to reduce miscarriage and infection risk).
If removal requires invasive procedure or threads not visible, leave the IUD in situ and counsel regarding increased risk of miscarriage and preterm birth.
If the woman does not wish to continue pregnancy, manage per abortion care guidance.
7. Ectopic Pregnancy
Even though the overall risk of ectopic pregnancy is lower in IUD users compared to those using no contraception, if a woman becomes pregnant with an IUD, about one-third to one-half of such pregnancies may be ectopic.
Presenting symptoms: amenorrhoea, abdominal pain, spotting.
Management is as per standard ectopic pregnancy protocols (medical or surgical).
8. Missing Threads
This is one of the most common reasons for review after IUD insertion.
Possible causes:
Spontaneous expulsion
Retraction into cervix or uterus
Breakage of threads
Perforation or embedment
Management:
Perform speculum examination; use cytology brush to locate threads in the endocervical canal.
If not visible, arrange pelvic ultrasound to confirm device position.
If intrauterine position is confirmed and the woman wishes to continue, no further action is needed until removal.
If malpositioned or perforated, manage as above.
9. Device Embedment or Fracture
Embedment occurs when the arms of the device become embedded in the myometrium, often discovered during difficult removal.
Fracture is rare and may occur if excessive traction is applied during removal or if the device has degraded with time.
Imaging (ultrasound or X-ray) should be performed to locate retained fragments.
Specialist removal under hysteroscopy may be required.
10. Other Rare Issues
Vasovagal syncope during insertion — usually self-limiting; patient should be placed supine and observed.
Expulsion unnoticed — counsel patients to check threads regularly.
Allergy or intolerance to copper — rare; device should be removed if systemic or local reaction occurs.
Patient Counselling Points
Explain that most complications are rare and manageable.
Emphasise the importance of checking threads after each menstrual period.
Advise to seek review for abdominal pain, heavy bleeding, fever, or foul discharge.
Reassure that IUDs do not increase long-term risk of infertility; fertility returns promptly after removal.
Suggested Exam Tips / Approach
- Be ready to quote key durations (e.g. 6 years, 8 years, “until menopause”)
- In a scenario question, always ensure pregnancy exclusion before insertion
- For “complication management” questions, always think about lost threads, suspected perforation, infection first
- In a “choice of method” scenario, weigh bleeding patterns, patient preference (hormonal vs non-hormonal), parity, breastfeeding status, and medical contraindications
- Use the guideline’s switching tables when asked: e.g. switching from CHC, injectable, POP, implant to IUD
- Keep in mind recent changes (e.g. extended use of LNG-IUD) for “current practice” questions
According to the UK Medical Eligibility Criteria (UKMEC 2016, adapted from WHO MEC),
Cu-IUCD use in women with prosthetic heart valves is Category 2 — meaning the advantages generally outweigh the theoretical or proven risks.
The main concern is the small risk of infective endocarditis during IUCD insertion.
Prophylactic antibiotics are not routinely recommended before insertion (RCOG, FSRH, 2019).
The procedure should be done under aseptic technique by an experienced provider.
Key takeaway:
Cu-IUCD can be safely used in women with Valvular and congenital heart disease/ prosthetic heart valves, provided:
There’s no active pelvic infection,
Aseptic technique is followed, and
Prophylaxis is considered only if the clinician deems the individual risk high.
Reference:
FSRH (Faculty of Sexual and Reproductive Healthcare) Clinical Guideline: Intrauterine Contraception (2023 update)
For FSRH Intrauterine Contraception Guideline 2023 click below 👇🏼
